Tesla launches virtual power plant in Puerto Rico, could become world’s biggest
Electrek
NOVEMBER 2, 2023
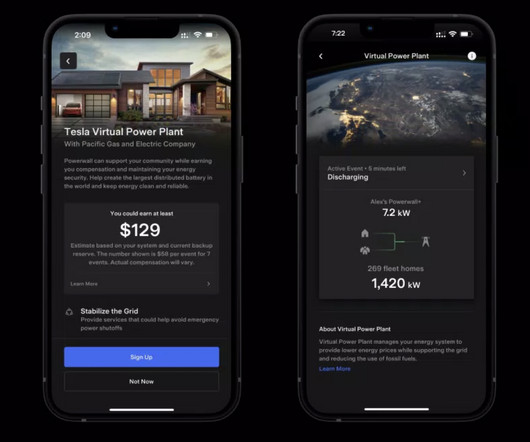
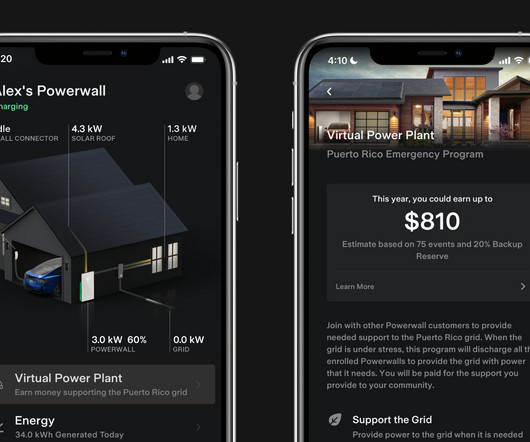
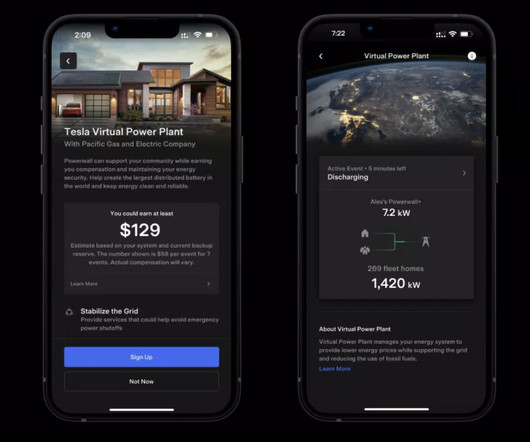
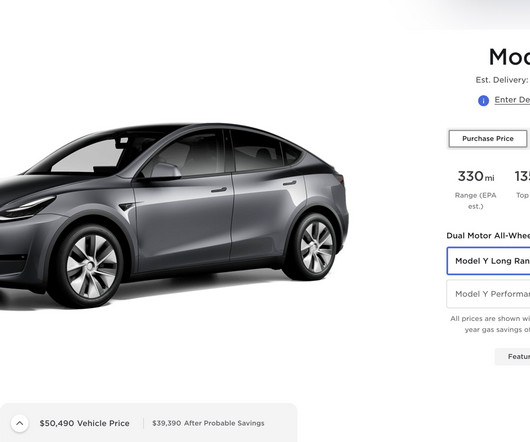
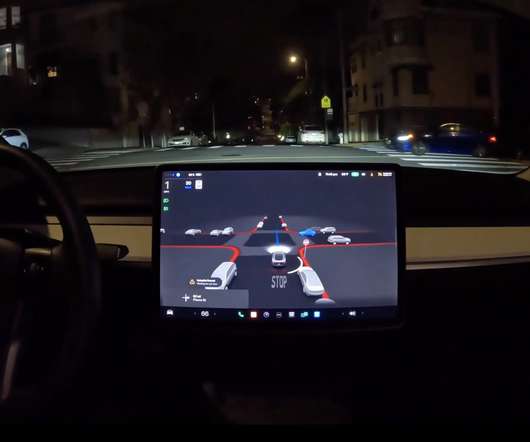
Tesla has officially launched its virtual power plant in Puerto Rico and it could quickly become the world’s biggest thanks to the large Powerwall user base in the region. more… The post Tesla launches virtual power plant in Puerto Rico, could become world’s biggest appeared first on Electrek.



































Let's personalize your content