Fraunhofer analysis finds battery-electric trucks have energy cost advantage over hydrogen trucks
Green Car Congress
FEBRUARY 8, 2022
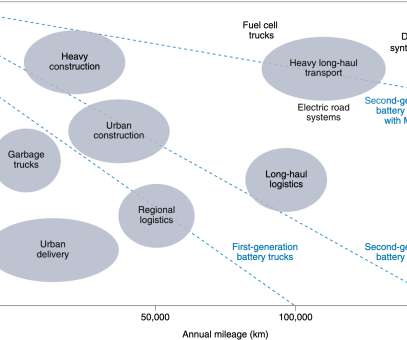
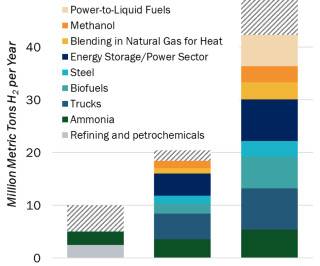
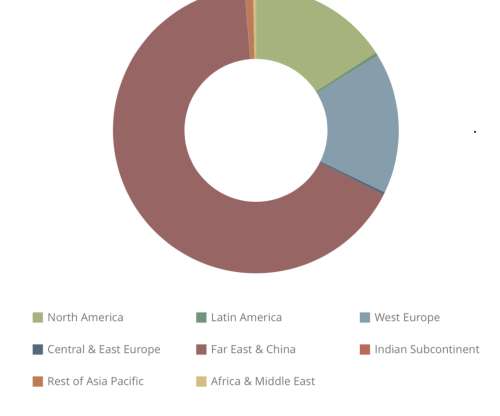
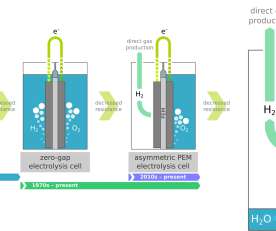
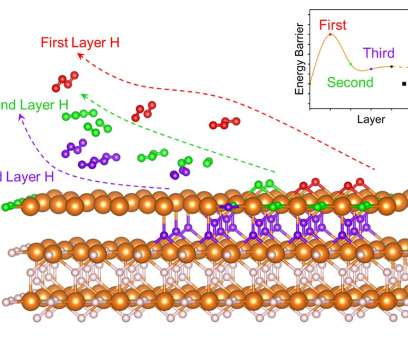
In a recent analysis, Fraunhofer ISI concludes that battery-electric drives will become established in heavy-duty applications in the commercial vehicle sector and that fuel cells will remain a niche application. The decisive factor for the switch to battery-electric vehicles is the energy cost advantage compared to hydrogen and diesel.




























Let's personalize your content